Accelerating the development with the attentive help of your needs.
[Our Business]
Nexredge accelerate the development of the research seeds of biological drugs and “human cell therapy and gene therapy products” based on biotechnology by supporting the process development and commercialization in accordance with the GMP guidelines and pharmaceutical regulations.
We offer comprehensive and highly specialized support in plant construction, development in manufacturing process and the machinery, building the organization, GMP and pharmaceutical applications.
Our goal is to provide a service that change the method of biopharmaceuticals development.
[Nexredge solution]
[Our Business]
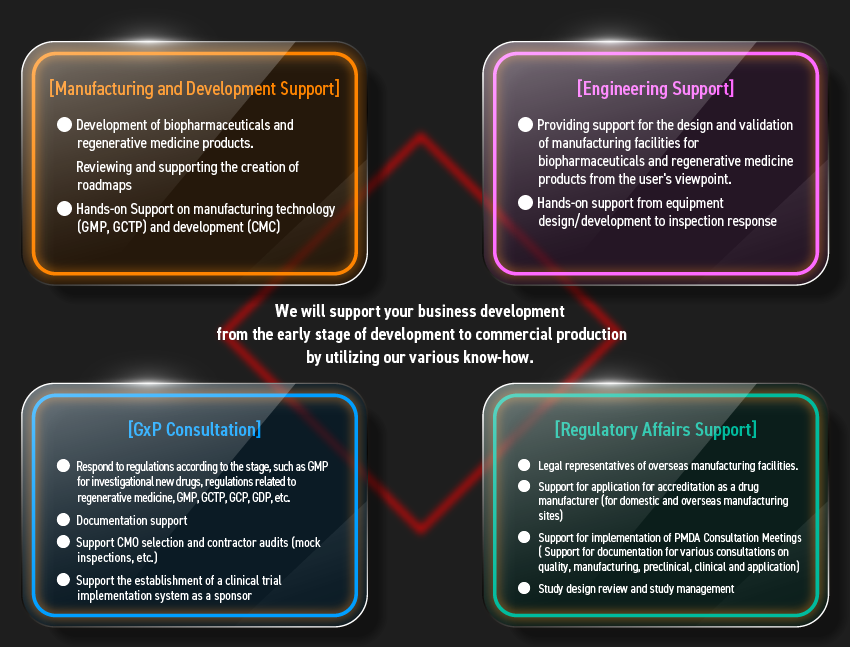
[Service Diagram]
※Mouse over any item to see its description.
Engineering
Support
Support
Development
Support
Support
Project
Management
Management
Regulatory
Consultation
Consultation
Development of Investigational New Drugs
TPP
(Target Product Profile)
(Target Product Profile)
Validation
Regulatory Consultation
supply of investigational new drugs
Project Management
Documentation
Employee Education / Training
Improvement of Manufacturing Process
Discussion of transportation service providers
Regulatory Affairs
Project Management
Regulatory Consultation
